Step 1: Find valence e- for all atoms. Add them together.
C=4x2=8
F=7x2=14
Total=22
Step2: Find octet e- for each atom and add them together.
C=8x2=16 F=8x2=16
Total=32
Step3: Subtract step 1 total from step 2. Gives you bonding e-.
32-22=10e-
Step 4: Find number of bonds by diving the number in step 3 by 2(because each bond is made of 2 e-)
10e-/2= 5 bond pairs
Step 5: Find the number of nonbonding (lone pairs) e-. Subtract step 3 number from step 1.
22-10= 12e-=6 lone pairs
Use information from step 4 and 5 to draw the lewis structure.
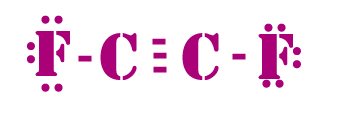
Lewis dot structure of C2F2
Alternatively a dot method can be used to draw the lewis structure of C2F2
Calculate the total valence electrons in the molecule.
C=4x2=8
F=7x2=14
Total=22
Put two canbon atoms in the center side by side.Put one fluorine on each carbon atom. Put the electrons and arrange them until all atoms get octet state.


